Are Serum Vitamin D Levels Associated With Dry Eye Disease? Results From the Study Group for Environmental Eye Disease
Article information
Abstract
Objectives
Dry eye disease (DED) is an increasingly important public health problem in Korea. Previous studies conducted in Korea have reported inconsistent results regarding the protective effects of vitamin D on DED, and these discrepancies may be related to the relatively simple questionnaire that has been used. Thus, we evaluated the association of serum vitamin D levels with DED using the ocular surface disease index (OSDI).
Methods
The present study evaluated data from participants in the Study Group for Environmental Eye Disease (2014-2015). This group included data from 752 participants, and data from 740 participants (253 men and 487 women) were analyzed in the present study. DED severity was evaluated using the OSDI.
Results
Higher serum vitamin D levels were associated with a non-significantly reduced risk of DED in the crude analysis (odds ratio [OR], 0.991; 95% confidence interval [CI], 0.971 to 1.011) and in the adjusted analysis (OR, 0.988; 95% CI, 0.966 to 1.010). In the crude analysis of no/mild DED vs. moderate/severe DED, men exhibited a decreased risk with increasing serum vitamin D levels (OR, 0.999; 95% CI, 0.950 to 1.051), while women exhibited an increased risk (OR, 1.003; 95% CI, 0.979 to 1.027). In these analyses, we found no significant associations.
Conclusions
The findings of the present study support previous reports that serum vitamin D levels are not associated with DED.
INTRODUCTION
Dry eye disease (DED) is a tear film disorder caused by chronic inflammation of the lacrimal functional unit [1]. DED is one of the most common complaints of patients who visit an ophthalmologist, and most patients with this condition experience symptoms including ocular discomfort, pruritus, redness, pain, and eye fatigue [1-3]. DED is also becoming an increasingly important public health problem because it affects vision-related quality of life (e.g., through adverse effects on reading and driving ability) [2-4]. Moreover, the prevalence of DED is increasing worldwide, and its prevalence is higher among Asian populations than among Western populations [5-10]. A Korean study reported that the prevalence of DED was 8.0% based on physician diagnoses and 14.4% based on DED symptoms [11].
Recent studies have suggested that vitamin D can protect against DED [12-15]. Vitamin D is a fat-soluble vitamin that is produced by cutaneous synthesis after sun exposure [16], and it has well-known musculoskeletal functions in the cartilage and bone [17,18]. Vitamin D has also been reported to have protective effects against various health problems, such as cardiovascular disease (CVD), diabetes, malignancies [19-22], and some eye disorders [23,24]. Some Korean studies have evaluated the associations of serum vitamin D levels with DED using Korea National Health and Nutrition Examination Survey (KNHANES) data, but their results were inconsistent [25-27]. They used a simple questionnaire about dry eye syndromes [28]. Therefore, we investigated the association between serum vitamin D levels and DED among Korean adults based on the ocular surface disease index (OSDI).
METHODS
The present study evaluated data from the Study Group for Environmental Eye Disease (SEED). The SEED study is an ancillary study of the Cardiovascular and Metabolic Disease Etiology Research Center (CMERC) cohort. The CMERC study is an ongoing cohort study (started in 2013) investigating risk factors for CVD and metabolic disease among the general population of healthy community-dwelling Korean adults (≥30 years old) [29]. The SEED study is an ongoing multidisciplinary study (started in 2013) that evaluates diagnostic and therapeutic biomarkers for environmental eye diseases. The present study analyzed data that were collected during 2014-2015 from the SEED study (752 participants), although we excluded 12 participants because they had missing data for relevant variables. Thus, we analyzed data from 740 participants (253 men and 487 women). The protocol of the present study was approved by the institutional review board of Severance Hospital (Yonsei University College of Medicine). All participants provided informed consent before participating in the CMERC and SEED studies.
In the present study, DED was defined as an OSDI score of ≥13.0. The OSDI uses a DED-specific questionnaire that was developed by the Outcomes Research Group at Allergan, and consists of questions regarding vision-related function (4 questions), eye symptoms (5 questions), and environmental triggers related to DED (3 questions). Each question is scored from 0 to 4, and the total OSDI score is calculated using the following formula: OSDI=([sum of scores for all questions answered]×100)/([total number of questions answered]×4), with results on a scale of 0 to 100. No DED (i.e., normal status) is defined as an OSDI score of <13.0. The severity of DED is defined as mild (scores of 13.0-22.9), moderate (23.0-32.9), and severe (scores of 33.0-100.0). Previous studies have confirmed that the OSDI is a valid and reliable tool for measuring DED symptoms and their effects on vision-related quality of life [30,31]. Trained interviewers individually administered the questionnaires to collect data regarding the participants’ demographic information, medical history, and OSDI.
Serum vitamin D levels (25-hydroxyvitamin D) were assessed using a chemiluminescence immunoassay (Liason, DiaSorin, Saluggia, Italy). Peripheral blood samples were obtained after an overnight fast, and all bioassays were performed at a central laboratory (Seoul Clinical Laboratories R&D Center, Seoul, Korea). Concentrations of blood glucose were measured using a colorimetric analyzer, while lipid profiles (including total cholesterol and high-density lipoprotein [HDL] cholesterol) were analyzed enzymatically. Glycated hemoglobin (HbA1c) concentrations were measured using high-performance liquid chromatography. Height was measured using a stadiometer (DS-102; Jenix, Seoul, Korea), and body weight was measured using a digital scale (DB-150; CAS, Seongnam, Korea). Body mass index (BMI) was calculated as body weight divided by height squared (kg/m2). Systolic and diastolic blood pressures were measured 3 times in the right arm, and the average values of the second and third measurements were used for the analysis [29].
Previous studies have revealed that the major risk factors or confounders for DED include age, women, BMI, smoking, history of eye surgery, diabetes mellitus, visual display terminal (VDT) use, contact lens use, and education level [10,32,33]. Thus, when available, we included these variables in our multivariate analyses. Diabetes mellitus was defined as a fasting blood glucose concentration of ≥126 mg/dL, an HbA1c concentration of ≥6.5%, or the current use of oral medication or insulin injection. Participants were classified according to smoking status as never smokers, former smokers, or current smokers. Because the effect sizes varied according to gender, we performed analyses of all participants together and of men and women separately. Continuous and normally distributed variables were compared using the t-test, and non-normally distributed continuous variables were compared using the Wilcoxon rank-sum test. Categorical variables were compared using the chi-square test. All tests were 2-tailed, and differences were considered statistically significant at p-values of <0.05. The associations of serum vitamin D levels with DED were evaluated using logistic regression analyses. Model 1 was not adjusted for any variables, model 2 was adjusted for age and BMI, and model 3 was adjusted for age, BMI, systolic/diastolic blood pressure, total cholesterol, HDL cholesterol, fasting glucose concentration, contact lens use, VDT use, history of eye surgery, and education level. The results of the logistic regression analyses were reported as odds ratios (ORs) with 95% confidence intervals (CIs). All analyses were performed using SAS version 9.4 (SAS Institute Inc., Cary, NC, USA).
RESULTS
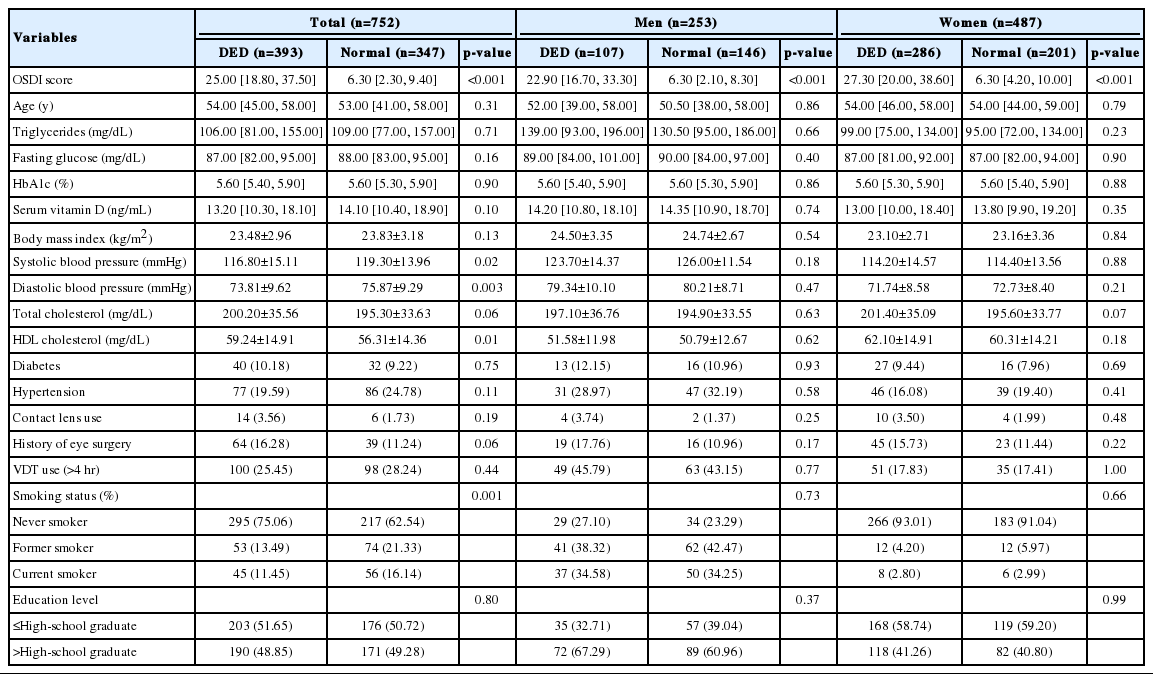
Table 1 shows the participants’ characteristics. As defined by an OSDI score of ≥13.0, the prevalence of DED among the 740 participants was 53.1%. The prevalence of DED among women was significantly higher than among men (58.7 vs. 42.3%, respectively; p<0.01). The median age was 53.0 years (51.0 years for men and 54.0 years for women). The univariate analyses revealed significant gender-related differences in the values for serum vitamin D, age, BMI, systolic blood pressure, diastolic blood pressure, HDL cholesterol, triglycerides, fasting glucose, history of hypertension, VDT use for >4 hours, smoking status, and education level. No gender-related differences were observed in the values for total cholesterol, HbA1c, history of diabetes mellitus, or contact lens use.
The participants’ characteristics according to DED status are shown in Table 2. Serum vitamin D levels were slightly higher among individuals without DED than among individuals with DED, and this non-significant association was observed among all participants (p=0.10), among men (p=0.74), and among women (p=0.35). The only significant differences in the overall analyses were observed in the values for systolic blood pressure, diastolic blood pressure, and HDL cholesterol among all participants.
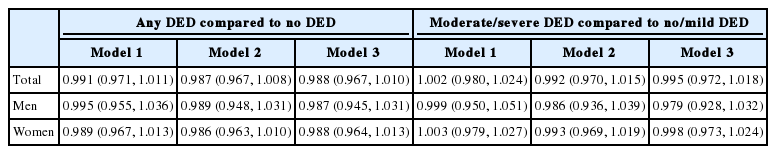
Higher serum vitamin D levels were associated with a non-significant reduction in the risk of DED in the crude analysis (OR, 0.991; 95% CI, 0.971 to 1.011) and in the fully adjusted analysis (OR, 0.988; 95% CI, 0.967 to 1.010) among all participants (Table 3). Among men, the crude OR was 0.995 (95% CI, 0.955 to 1.036) and the adjusted OR was 0.987 (95% CI, 0.945 to 1.031). Among women, the crude OR was 0.989 (95% CI, 0.967 to 1.013) and the adjusted OR was 0.988 (95% CI, 0.964 to 1.013).
When we compared individuals with no/mild DED to individuals with moderate/severe DED, we observed that higher serum vitamin D levels were associated with a non-significant increase in the risk of moderate/severe DED in the crude analysis (OR, 1.002; 95% CI, 0.980 to 1.024) and a non-significant decrease in the fully adjusted analysis (OR, 0.995; 95% CI, 0.972 to 1.018). We found a non-significant decrease in the risk of moderate/severe DED among men (OR, 0.999; 95% CI, 0.950 to 1.051) and a non-significant increase in the risk among women (OR, 1.003; 95% CI, 0.979 to 1.027) in the crude analysis. These associations remained non-significant after adjusting for all covariates. In the adjusted analysis, the OR among women was 0.998 (95% CI, 0.973 to 1.024) (Table 3).
DISCUSSION
The prevalence of DED in the present study was 53.1% (42.3% among men and 58.7% among women). When we considered only moderate/severe DED, the prevalence was 30.3% (19.8% among men and 35.7% among women). Previous studies have revealed that the prevalence of DED has risen from 4.3 to 73.5% [5], and our results are within this range. We suggest that the diagnosis of mild DED could cause the prevalence of DED to be overestimated.
In addition, we observed various gender-related differences in the participants’ characteristics, and similar results have been observed in a previous study [26]. Some researchers have reported findings that may explain the gender-related differences, which could be related to differences in sex hormones or sunlight exposure times between men and women [26,34]. In the present study, our findings may suggest that women are more sensitive to the symptoms of DED. When comparing individuals with no DED to those with DED of any severity, the protective effect of vitamin D was <1.00, whereas this effect was >1.00 when individuals with no/mild DED were compared to those with moderate/severe DED. This may have been because women were more sensitive to the symptoms. When we compared participants with no/mild DED to those with moderate/severe DED in the crude analyses, we observed opposite directions of the associations of DED with elevated serum vitamin D levels among men and women.
The results of some studies have suggested that vitamin D levels can affect the immune system, and studies in mice have shown that vitamin D enhanced corneal epithelial barrier function through tight or gap junctions [12,13,35]. Moreover, vitamin D can control ocular surface inflammation by inhibiting Langerhans cell migration and corneal neovascularization [36,37]. In humans, researchers have reported that vitamin D supplementation is an effective and useful treatment for patients with DED [38]. Our results suggest that oral vitamin D supplementation is less likely to be effective than topical supplementation.
In our study, no association was found between serum vitamin D levels and DED. One possible explanation for this is that it may be difficult for serum vitamin D to reach the cornea because of its lack of vasculature. Another possibility is a positive relationship between aging and serum vitamin D levels. A previous study reported that the aged in Korea and Thailand had higher serum vitamin D levels than the young. They suggested that the elderly had more free time for outdoor activities and tended to have outdoor jobs, such as being farmers or fishermen. Moreover, younger people may use more sunblock than the elderly [26,39,40].
Previous studies conducted in Korea have evaluated the association of serum vitamin D with DED using KNHANES data, which were obtained using the participants’ responses to a questionnaire (“Have you ever been diagnosed with DED by an ophthalmologist?” and “Have you experienced symptoms of DED, such as dryness or irritation?”) [28]. However, these studies have reported inconsistent findings, as some researchers have defined DED based on an ophthalmological diagnosis, while others considered both an ophthalmological diagnosis and DED symptoms. For example, Jee et al. [26] reported that the risk of DED was lower in higher serum vitamin D quintiles (quintiles 5 vs. 1: OR, 0.85; 95% CI, 0.55 to 1.30; p for trend=0.08), although this association was not statistically significant. Yoon et al. [25] also reported that low serum vitamin D levels were a risk factor for DED (OR, 1.158; 95% CI, 1.026 to 1.308), and Kim et al. [27] reported an elevated crude risk of DED at low serum vitamin D levels (OR, 1.61; 95% CI, 1.00 to 2.61; p=0.04), although this association was attenuated after adjusting for covariates (OR, 1.42; 95% CI, 0.84 to 2.40). However, the rate of DED diagnosis is relatively low, and its prevalence could be underestimated if it is based on the ophthalmological diagnosis rate, or overestimated if it is based on DED symptoms, which are shared with other eye diseases.
The present study used OSDI scores to identify cases of DED, as this tool is valid and reliable for measuring DED symptoms and their effects on vision-related quality of life [30,31]. We also compared individuals with and without DED, or with no/mild DED versus moderate/severe DED, because mild symptoms may lead to a misclassification of DED. These analyses failed to detect a significant association between serum vitamin D levels and DED, and our findings support previous reports of no significant association between these variables. This absence of a significant association may be related to the possibility that serum vitamin D levels do not reflect the levels in lacrimal fluid. It is also possible that species-based or racial differences affect these associations, as previous studies regarding the protective effect of vitamin D were conducted in mouse models [12,13,35].
The present study has some limitations that warrant consideration. First, the cross-sectional design precludes any conclusions regarding causality, although previous studies conducted in Korea have also used cross-sectional designs. Second, our dataset was relatively limited compared to the KNHANES data set, which may limit the representativeness of our findings. Nevertheless, our use of the OSDI to detect DED likely provided more detailed and objective information regarding DED severity. Third, we did not consider seasonal variation in serum vitamin D levels. To deal with this variation, previous studies using KNHANES data adjusted for sunlight exposure or occupation. In the KNHANES, information about sunlight exposure and occupation was acquired through the questionnaire, and the surveys were conducted throughout the year. Our data were gathered from October 2014 to January 2015 and from October 2015 to December 2015. Thus, our data are likely to contain less seasonal variation than KNHANES.
The present study provides epidemiological data regarding the absence of an association of serum vitamin D levels and DED in the Korean general population. This result is consistent with the findings of Jee et al. [26], who analyzed KNHANES data. Further studies are needed to investigate the mechanisms and management of DED.
ACKNOWLEDGEMENTS
This study was funded by the Korean Health Technology R&D Project (HI13C0055).
Notes
CONFLICT OF INTEREST
The authors have no conflicts of interest associated with the material presented in this paper.